 Multiple Choice Questions
Multiple Choice QuestionsWhen NaCl is dissolved in water , the sodium ions becomes :
oxidised
reduced
hydrolysed
hydrated
Which of the following is used as moderator :
Hard water
Heavy water
Non-ionised water
Mineral water
When SO2 gas is passed through an acidified K2Cr2O7 solution :
the solution becomes blue
SO2 gets reduced
Cr(SO4)3 is formed
the solution becomes colourless
In the following reaction ,
P4 + 3NaOH + 3H2O PH3 + 3NaH2PO2
phosphorus is oxidised
phosphorus is oxidised and reduced
phosphorus is reduced
sodium is oxidised
B.
phosphorus is oxidised and reduced
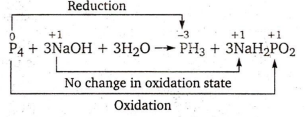
Hence, in this reaction oxidation and reduction of phosphorus takes place and there occurs no change in the oxidation state of sodium .
The temporary hardness of water due to calcium bicarbonate can be removed by adding :
CaCO3
Ca(OH)2
CaCl2
HCl
The substance used in smoke screen is :
sodium chloride
sodium phosphate
calcium fluoride
calcium phosphide
Which of the following complexes do not follow EAN rule ?
[Fe(CN)6]4-
[Fe(CN)6]3-
Ni(CO)4
[PdCl4]2-
