 Multiple Choice Questions
Multiple Choice QuestionsWhen NaCl is dissolved in water , the sodium ions becomes :
oxidised
reduced
hydrolysed
hydrated
Which of the following is used as moderator :
Hard water
Heavy water
Non-ionised water
Mineral water
When SO2 gas is passed through an acidified K2Cr2O7 solution :
the solution becomes blue
SO2 gets reduced
Cr(SO4)3 is formed
the solution becomes colourless
In the following reaction ,
P4 + 3NaOH + 3H2O PH3 + 3NaH2PO2
phosphorus is oxidised
phosphorus is oxidised and reduced
phosphorus is reduced
sodium is oxidised
Which of the following will show aromatic behaviour ?
![]()
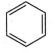
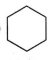
None of these
B.

Aromatic compounds exhibits following properties :
(i) They obey Huckel (4n + 2) e- rule .
(ii) They involve delocalisation of -electrons .
(iii) Molecule must be planar (ie , sp2 hybridised) .
Benzene satisfy all these conditions , so it will be aromatic in nature .
The temporary hardness of water due to calcium bicarbonate can be removed by adding :
CaCO3
Ca(OH)2
CaCl2
HCl
The substance used in smoke screen is :
sodium chloride
sodium phosphate
calcium fluoride
calcium phosphide
Which of the following complexes do not follow EAN rule ?
[Fe(CN)6]4-
[Fe(CN)6]3-
Ni(CO)4
[PdCl4]2-
