Explain the following giving an appropriate reason in each case.
Structures of Xenon fluorides cannot be explained by Valence Bond approach.
According to the valence bond approach, covalent bonds are formed by the overlapping of the half-filled atomic orbital. But xenon has a fully filled electronic configuration. Hence the structure of xenon fluorides cannot be explained by VBT.
For example: In case of XeF2
Hybridization is:
Total, valence electron =8 for Xe
Monovalent atom F=2
So, total hybrid orbitals =1/2 [valence electron + monovalent atom + cation + anion] as no cation and anion so term is zero
So hybrid orbital are ½[8+2+0+0] = 5
So hybridization = sp3d
According to sp3d hybridization, the structure should be Trigonal bipyramidal, but the actual structure is linear. So, VBT fails to answer this however VSPER theory explains the liner shape.
How would you account for the following?
i) Many of the transition elements are known to form interstitial compounds.
ii)The metallic radii of the third (5d) series of transition metals are virtually the same as those of the corresponding group member of the second (4d) series.
iii) Lanthanoids from primarily +3 ions, while the actinoids usually have higher oxidation states in their compounds, +4 or even + 6 being typical.
(i) Formation of the interstitial compounds: Transition elements exist in CCP and HCP structure which are known to possess vacant position (Holes),such that transition elements form a few interstitial compounds with elements possessing small atomic radii, like hydrogen, carbon boron, and nitrogen. Small atoms of these type of elements get entrapped in between the void spaces (called as interstices) of the metal lattice. Some of the characteristics of the interstitial compound. These compounds show basically the same chemical properties as the parent metals but vary in the physical properties such as density and hardness. For example, the best known is tungsten carbide WC that is extremely hard and used in steel cutting tools, armour and jewellery.
Explanation: Interstitial compounds are hard and dense in nature. This is because; the smaller atoms of the lighter elements occupy the interstices in the lattice, leading to the much closely packed structure. Because of greater electronic interactions, the strength of metallic bonds also increases.
(ii) Metallic radii of third (5d) series of transition metals are virtually same as those of (4d) series because of the lanthanoid contraction.
Lanthanoid contraction: it is the phenomenon occurring in the 3rd series of the transition element. As they have electron filling up the 4f shell which is filled with the 5d series. The 4f shell has very weak shielding effect and is highly attracted towards the Nucleus due to high nuclear charge so the filling of 4f orbitals before 5d orbital resulting in their atomic radius which is very similar to the elements of the 2nd series i.e. Zr=160 atomic radius and Hf=159 atomic radius.
(iii) The wide range of oxidation states of actinoids is attributed to the fact that the 5f, 6d and 7senergy levels are comparable energies. Therefore all these three sub shells can participate. But the most common oxidation state of actinoids is +3.
Assign reasons for the following:
(i) Copper (I) ion is not known in aqueous solution.
(ii) Actinoids exhibit greater range of oxidation states than Lanthanoids.
Stability in the aqueous medium depends on the hydration energy of ions when they attract to water to water molecules. In aqueous solution, Cu+ ion undergoes oxidises and reduces simultaneously
in aqueous to give Cu and Cu2+ ion.

The relative stability of different oxidation states can be seen from their
Electrode potentials 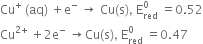
Due to more reduction electrode potential value of Cu+, it undergoes oxidation reaction quite feasible. Hence, the copper (I) ion is not known in aqueous solution.
(ii) The actinoids show a larger number of oxidation states because of the very small energy gap between the 5f, 6d and 7s subshells. The energies are decided on the basis of the (n+1) rule.
The (n+1) values of the three orbitals are under:
5f=5+3=8
6d=5+2=8
7s=7+ 1=8
All of the value comes to be same. Hence they have the same energy.
