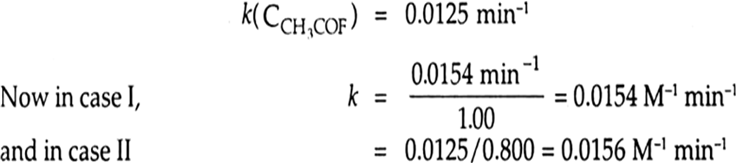We have given that
Substituting these values in the equation
we get
or
|
Run |
Initial |
Initial |
Initial rate (mol s–1) |
|
concentration |
concentration |
||
|
1 |
0.10 M |
0.1 M |
2.1 x 10–3 |
|
2 |
0.20 M |
1.0 M |
8.4 x 10–3 |
|
3 |
0.20 M |
2.0 M |
8.4 x 10–3 |
Determine the order of reaction with respect to A and with respect to B and the overall order of reaction.
Order with respect to A = 2.
Order with respet to B = 0.
Overall order of reaction = 2.
D.
increase in number of molecular collisions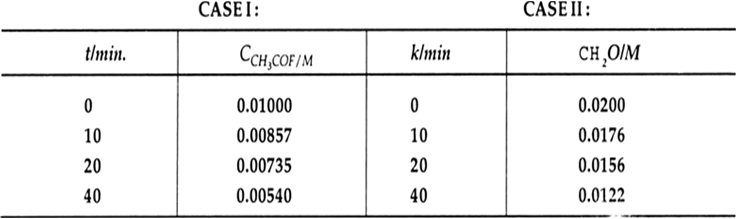
Let rate k(CCH3COF)a (CH2O)b
CH2O >> CCH3COF and in the second case, CH2O << CCH3COF. In the first case we determine the order of the reaction with respect to CH3 COF. We note that the reaction is not of zero order as rate of reaction changes with time.
Therefore k(CH2O) = 0.0154 min–1 and we note the order of reaction with respect to CH3COF is 1.
Now we determine the order of raction with respect to water.
Again,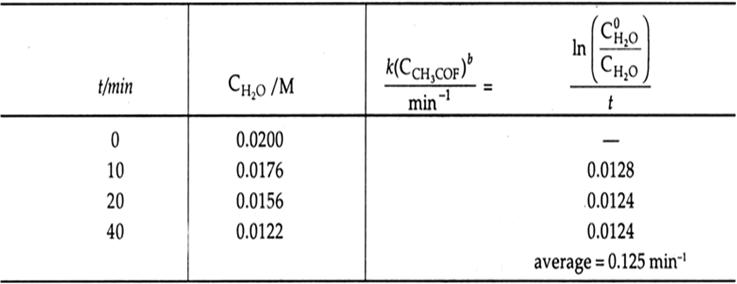
The reaction is first order in H2O and we have