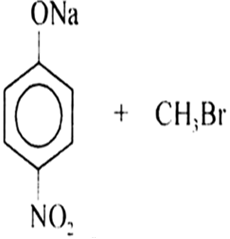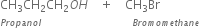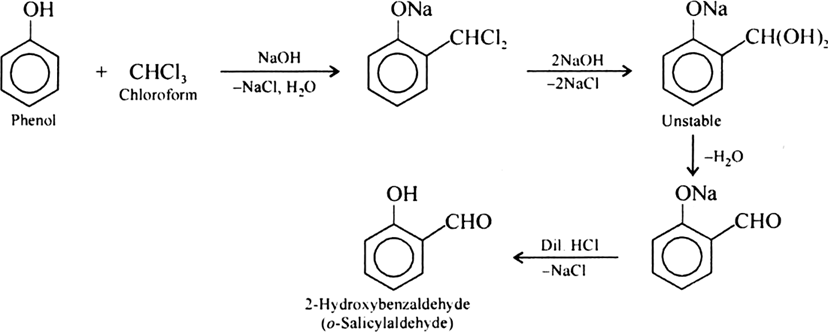
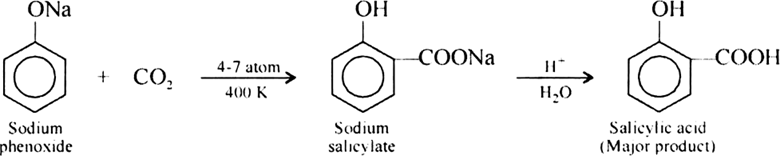
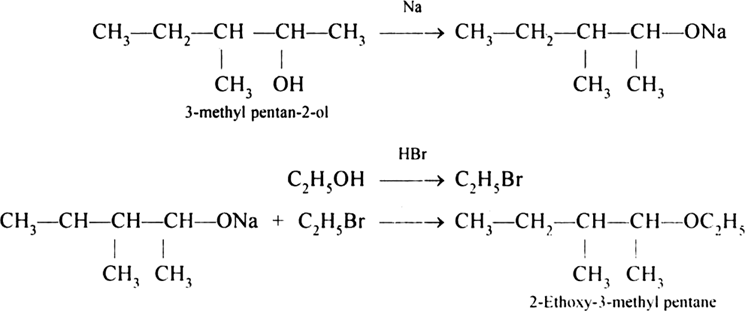
It is an important laboratory method for the preparation of symmetrical and unsymmetrical ethers. In this method, an alkyl halide is allowed to react with sodium alkoxide.
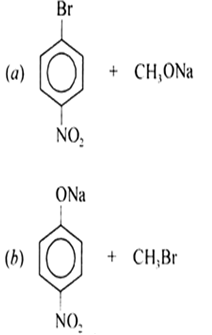
In structure (a), group sodium methoxide (CH3ONa) is a strong nucleophile also it is a strong base. Hence, an elimination reaction predominates over a substitution reaction.