The rate of reaction can be express by Dividing the individual rate expression by the coefficients in the balanced chemical equation.
(a) Show graphically how the rate of first order reaction vary with only one reactant depends concentration of reactant.
(b) Give one example of first order reaction.
(a)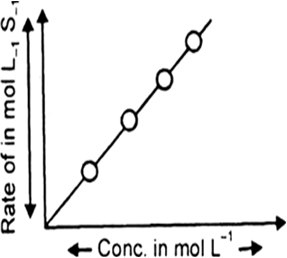
(b) Example of first order reaction is![]()
