 Short Answer Type
Short Answer TypeAlthough chlorine is an electron withdrawing group, yet it is ortho-, Para-directing in electrophilic aromatic substitution reactions. Explain why it is so?
Chlorine withdraws electrons through inductive effect and releases electrons through resonance. Through inductive effect, chlorine destabilises the intermediate carbocation formed during the Electrophilic substitution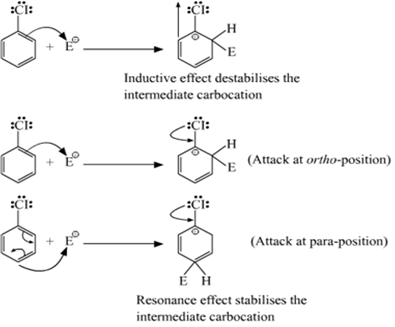
Through resonance, halogen tends to stabilise the carbocation and the effect is more pronounced at ortho-and Para-position. The inductive effect is stronger than resonance and causes net electron withdrawal and thus causes net deactivation. The resonance effect tends to oppose the inductive effect for the attack at ortho-and Para-position and hence makes the deactivation less for ortho- and Para-attack. Reactivity is thus controlled, by the stronger inductive effect and orientation is controlled by a resonance effect.
Draw the structure and name the product formed if the following alcohols are oxidised. Assume that an excess of oxidising agent is used.
(i) CH3CH2CH2CH2OH
(ii) 2-butenol
(iii) 2-methyl-1-proponal
Write chemical equation for the following conversions:
(i) Nitrobenzene to benzoic acid.
(ii) Benzyl chloride to 2-phenylethanamine.
(iii) Aniline to benzyl alcohol.
What are the following substances? Give one example of each one of them.
(i) Tranquilizers
(ii) Food preservatives
(iii) Synthetic detergents
Illustrate the following name reaction giving suitable example in each case:
(i) Clemmensen reduction
(ii) Hell-Volhard-Zelinsky reaction
Give simple tests to distinguish between the following pairs of compounds.
(i) Pentan-2-one and Pentan-3-one
(ii) Benzaldehyde and Acetophenone
(iii) Phenol and Benzoic acid
Illustrate the following reactions giving a suitable example for each.
(i) Cross aldol condensation
(ii) Decarboxylation
Give simple tests to distinguish between the following pairs of compounds
(i) Pentan-2-one and Pentan-3-one
(ii) Benzaldehyde and Acetophenone
(iii) Phenol and Benzoic acid
