 Multiple Choice Questions
Multiple Choice QuestionsFor a first order reaction, (A) → products, the concentration of A changes from 0.1 M to 0.025 M in 40 minutes. The rate of reaction when the concentration of A is 0.01 M is
1.73 x 10–5 M/ min
3.47 x 10–4 M/min
3.47 x 10–5 M/min
3.47 x 10–5 M/min
B.
3.47 x 10–4 M/min
By first order kinetic rate constant,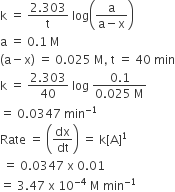
Iron exhibits + 2 and +3 oxidation states. Which of the following statements about iron is incorrect?
Ferrous oxide is more basic in nature than the ferric oxide.
Ferrous compounds are relatively more ionic than the corresponding ferric compounds.
Ferrous compounds are less volatile than the corresponding ferric compounds.
Ferrous compounds are less volatile than the corresponding ferric compounds.
Kf for water is 1.86K kg mol–1.If your automobile radiator holds 1.0 kg of water, how many grams of ethylene glycol (C2H6O2) must you add to get the freezing point of the solution lowered to –2.8°C?
72 g
93 g
39 g
39 g
Which of the following compounds can be detected by Molisch’s test?
Nitro compounds
Sugars
Amines
Amines
Aspirin is known as
acetyl salicylic acid
phenyl salicylate
acetyl salicylate
acetyl salicylate
Ortho–Nitrophenol is less soluble in water than p– and m– Nitrophenols because
o–Nitrophenol is more volatile in steam than those of m – and p–isomers
o–Nitrophenol shows Intramolecular H–bonding
o–Nitrophenol shows Intermolecular H–bonding
o–Nitrophenol shows Intermolecular H–bonding
What is DDT among the following
Greenhouse gas
A fertilizer
Biodegradable pollutant
Biodegradable pollutant
Iodoform can be prepared from all except
ethyl methyl ketone
isopropyl alcohol
3-methyl-2-butanone
3-methyl-2-butanone
In the given transformation, which of the following is the most appropriate reagent?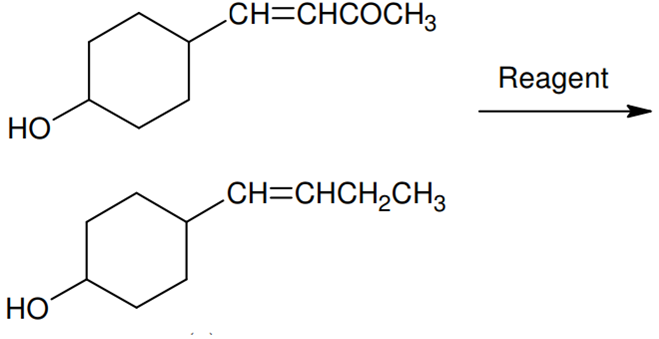
NH2NH2,O-H
Zn − Hg/HCl
Na,Liq.NH3
Na,Liq.NH3
