 Multiple Choice Questions
Multiple Choice QuestionsThe equation which is balanced and represents the correct product(s) is
Li2O + 2KCl → 2LiCl + K2O
[COCl(NH3)5]+ + 5H+ → Co2+
[Mg(H2O)6]2+ + (EDTA)4-  [Mg(EDTA)]2+ + 6H2O
[Mg(EDTA)]2+ + 6H2O
[Mg(H2O)6]2+ + (EDTA)4-  [Mg(EDTA)]2+ + 6H2O
[Mg(EDTA)]2+ + 6H2O
B.
[COCl(NH3)5]+ + 5H+ → Co2+
In SN2 reactions, the correct order of reactivity for the following compounds CH3Cl, CH3CH2Cl, (CH3)2CHCl and (CH3)3CCl is
CH3Cl > (CH3)2CHCl >CH3CH2Cl > (CH3)3CCl
CH3Cl > CH3CH2Cl > (CH3)2CHCl >(CH3)3CCl
CH3CH2Cl > CH3Cl >(CH3)2CHCl >(CH3)3CCl
CH3CH2Cl > CH3Cl >(CH3)2CHCl >(CH3)3CCl
On heating an aliphatic primary amine with chloroform and ethanolic potassium hydroxide, the organic compound formed is
an alkanol
an alkaneodiol
an alkyl cyanide
an alkyl cyanide
The most suitable reagent for the conversion of
R-CH2 -OH →R- CHO is
KMnO4
K2Cr2O7
CrO3
CrO3
The major organic compound formed by the reaction of 1,1,1-trichloroethane with silver powder is
acetylene
ethene
2-butyne
2-butyne
Which one of the following bases is not present in DNA?
Quinoline
Adenine
Cytosine
Cytosine
Sodium phenoxide when heated with CO2 under pressure at 125oC yields a product which on acetylation products C.![]()
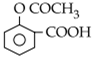


Considering the basic strength of amines in aqueous solution, which one has the smallest pKb value?
(CH3)2NH
CH3NH2
(CH3)3N
(CH3)3N
