 Short Answer Type
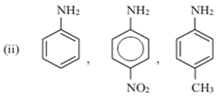
Short Answer TypeArrange the following in increasing order of their basic strength:
(i) C6H5 – NH2, C6H5 – CH2 – NH2, C6H5 – NH – CH3
Give reasons for the following:
(i) N2 is less reactive at room temperature.
(ii) H2Te is the strongest reducing agent amongst all the hydrides of Group 16 elements.
(iii) Helium is used in diving apparatus as a diluent for oxygen.
Give reasons for the following:
(i) Oxygen is a gas but sulphur is solid.
(ii) O3 acts as a powerful oxidising agent.
(iii) BiH3 is the strongest reducing agent amongst all the hydrides of Group 15 elements.
Explain the following facts giving appropriate reason in each case:
(i) NF3 is an exothermic compound whereas NCl3 is not.
(ii) All the bonds in SF4 are not equivalent.
 Long Answer Type
Long Answer Type(a) Draw the molecular structure of the following compounds.
(i) N2O5
(ii) XeOF4
(b) Explain the following observation:
(i) Sulphur has a greater tendency for catenation than oxygen.
(ii) ICI is more reactive than I2.
(iii) Despite the lower value of its electron gain enthalpy with a negative sign, fluorine (F2) is a stronger oxidizing agent than Cl2.
(a) Complete the following chemical equation
(i) Cu + HNO3 (dilute) --->
(ii) XeF4 + O2F2 -->
(b) Explain the following observation:
(i) Phosphorus has a greater tendency for catenation than nitrogen.
(ii) Oxygen is a gas but sulphur a solid.
(iii) The halogens are coloured. Why?
(i) 3Cu + 8HNO3(dilute) ----> 3Cu (No3)2 + 2NO + 4H2O
(ii) XeF4 + O2F2 ---> XeF6 + O2
(b)
(i)Catenation is the property of atoms of an element to link together and in case of nitrogen the size of it being small and due to electron repulsion it can exist stably in diatomic form
Catenation is much more common in phosphorous compounds than in nitrogen compounds. This is because of the relative weakness of the N-N single bond as compared to the P-P single bond. Since nitrogen atom is smaller, there is greater repulsion of electron density of two nitrogen atoms, thereby weakening the N-N single bond.
(ii) Oxygen forms O2 which is a gas and sulphur forms S8 which is solid this can be explained as:
Due to the small size of oxygen, it has less tendency for catenation and the high tendency of pp-pp multiple bonds, hence forms stable O2 molecules whereas sulphur because of its higher tendency for catenation and lesser tendency to form pp-pp multiple bonds forms S8 molecules having 8-membered puckered ring. Held together by strong covalent bonds and exist as polyatomic molecule, so it exists solid
(iii) All the halogens possess a valence shell electronic configuration s2, p5. This means that they contain unpaired electrons in their outermost p- orbital. These electrons absorb light and get promoted to higher orbitals. When they return to their ground state, they emit radiation which falls in the visible region of electromagnetic spectrum. Hence appear.
(a) Account for the following:
(i)Ozone is thermodynamically unstable.
(ii)Solid PCl5 is ionic in nature.
(iii)Fluorine forms only one oxoacid HOF.
(b) Draw the structure of
(i) BrF5
(ii) XeF4
OR
(i)Compare the oxidizing action of F2 and Cl2 by considering parameters such as bond dissociation enthalpy, electron gain enthalpy and hydration enthalpy.
(ii)Write the conditions to maximize the yield of H2SO4 by contact process.
(iii)Arrange the following in the increasing order of property mentioned:
(a)H3PO3, H3PO4, H3PO2 (Reducing character)
(b)NH3, PH3, AsH3, SbH3, BiH3 (Base strength)
