 Multiple Choice Questions
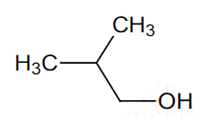
Multiple Choice QuestionsAmong the following the one that gives positive iodoform test upon reaction with I2 and NaOH is
CH3CH2CH(OH)CH2CH3
C6H5CH2CH2OH

The correct order of increasing acid strength of the compounds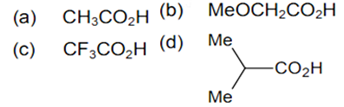
b < d < a < c
d < a < c < b
d < a < b < c
d < a < b < c
The best reagent to convert pent -3- en-2-ol into pent -3-en-2-one is
Acidic permanganate
Acidic dichromate
Chromic anhydride in glacial acetic acid
Chromic anhydride in glacial acetic acid
Reaction of cyclohexanone with dimethylamine in the presence of catalytic amount of an acid forms a compound if water during the reaction is continuously removed. The compound formed is generally known as
a Schiff’s base
an enamine
an imine
an imine
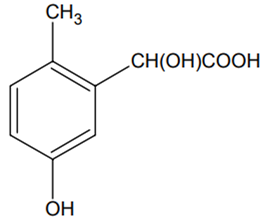
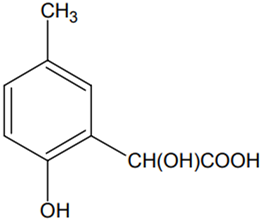
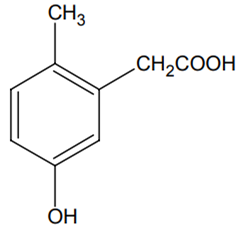
p-cresol reacts with chloroform in the alkaline medium to give the compound A which adds hydrogen cyanide to form, the compound B. The latter on acidic hydrolysis gives chiral carboxylic acid. The structure of the carboxylic acid is

Which of the following salts is the most basic in aqueous solution?
Pb(CH3COO)2
Al(CN)3
CH3COOK
FeCl3
Amongst the following compounds, the one that wil not respond to Cannizzaro reaction upon treatment with alkali as
Cl3CCHO
Me3CCHO
C6H5CHO
HCHO
A.
Cl3CCHO
For a carbonyl group to give Cannizaro reaction, it should not have an -hydrogen atom. Among the given compound Me3CCHO, C6H5CHO and HCHO respond to Cannizaro reaction but Cl3CCHO do not due to following reason.

When hydroxyl group attacks the carbonyl group a tetrahedral intermediate forms. This tetrahedral intermediate will revert to a carbonyl compound by expelling the best leaving group. The carbonium is resonance stabilised therefore it is better leaving group than OH. These initial products exchange a proton to give ion of carboxylic acid and trichloromethane (CHCl3).
Amongst the following compounds, the one which would not respond to iodoform test is
CH3CH(OH)CH2CH3
ICH2COCH2CH3
CH3COOH
CH3CHO
Which of the following reaction will not result in the formation of carbon - carbon bonds?
Cannizzaro reaction
Wurtz reaction
Reimer-Tiemann reaction
Friedel-Crafts acylation
