 Short Answer Type
Short Answer TypeArrange the following compounds in an increasing order of their solubility in water:
C6H5NH2, (C2H5)2NH, C2H5NH2Explain the following giving one example for each:
(i) Reimer-Tiemann reaction.
(ii) Friedel Craft’s acetylation of anisole.
i) Reimer-Tiemann Reaction: Reimer-Tiemann reaction involves the treatment of phenol with chloroform in the presence of aqueous sodium hydroxide at 340 K followed by hydrolysis of the resulting product to give 2-hydroxybenzaldehyde (salicylaldehyde). The chemical reaction can be represented as follows. 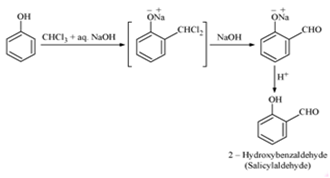
ii) Friedel-Crafts acetylation of anisole: Friedel-Crafts acetylation of anisole involves the treatment of anisole with either acetyl chloride or acetic anhydride to give 2-methoxyacetophenone (as a minor product) and 4-methoxyacetophenone (as a major product).
The chemical reaction can be represented as follows. 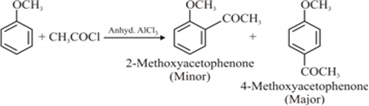
How would you obtain
(i) Picric acid (2, 4, 6-trinitrophenol) from phenol,
(ii) 2-Methylpropene from 2-methylpropanol?
What is essentially the difference between α-form of glucose and β-form of glucose? Explain.
 Long Answer Type
Long Answer Type(a) Differentiate between molarity and molality for a solution. How does a change in temperature influence their values?
(b) Calculate the freezing point of an aqueous solution containing 10.50 g of MgBr2in 200 g of water. (Molar mass of MgBr2 = 184 g) (Kf for water = 1.86 K kg mol-1)
