 Multiple Choice Questions
Multiple Choice QuestionsThe species which can best serve as an initiator for the cationic polymerization is
LiAlH4
HNO3
AlCl3
AlCl3
The standard reduction potentials for Zn2+/ Zn, Ni2+/ Ni, and F2+/ Fe are –0.76, –0.23 and –0.44 V respectively. The reaction X + Y2+ → X 2+ + Y will be spontaneous when
X = Ni, Y = Fe
X = Ni, Y = Zn
X =Fe, Y= Zn
X =Fe, Y= Zn
The equilibrium constant (Kc) for the reaction, N2(g) + O2 (g) → 2NO (g) at temperature T is 4 x 10-4. The value of Kc for the reaction 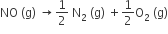 at the same temperature is
at the same temperature is
0.02
2.5 x 102
4 x 10-4
4 x 10-4
The electrons identified by quantum numbers n and l:
(a) n = 4, l = 1 (b) n = 4, l = 0 (c) n = 3, l = 2 (d) n = 3 , l = 1
Can be placed in order of increasing energy as
(c) < (d) < (b) < (a)
(d) < (b) < (c) < (a)
(b) < (d) < (a) < (c)
(b) < (d) < (a) < (c)
The pH of a 0.1 molar solution of the acid HQ is 3. The value of the ionisation constant, Ka of this acid is
3 x 10–1
1 x 10–3
1 x 10—5
1 x 10—5
The increasing order of the ionic radii of the given isoelectronic species is
Cl-, Ca2+, K+, S2-
S2-, Cl-, Ca2+,K+
Ca2+, K+, Cl-, S2-
Ca2+, K+, Cl-, S2-
In which of the following pairs the two species are not isostructural?
CO32- and NO3-
PCl+4 and SiCl4
PF5 and BrF5
PF5 and BrF5
Which of the following on thermal decomposition yields a basic as well as an acidic oxide?
NaNO3
KClO3
CaCO3
CaCO3