 Multiple Choice Questions
Multiple Choice QuestionsThe value of H for cooling 2 mole of an ideal monoatomic gas from 225C to 125C at constant pressure will be [ given Cp= ].
250 R
-500 R
500R
-250 R
The system that contains the maximum number of atoms is
4.25 g of NH3
8 g of O2
2g of H2
4g of He
The volume of ethyl alcohol ( density 1.15 g/cc) that has to be added to prepare 100 cc of 0.5 M ethyl alcohol solution in water is
1.15 cc
2 cc
2.15 cc
2.30 cc
The bond angle in NF3 (102.3) is smaller than NH4 (107.2). This is because of
large size of F compared to H
large size of N compared to F
opposite polarity of N in the two molecules
small size of H compared to N
C.
opposite polarity of N in the two molecules
In NF3 N is less electronegative as compared to F but in NH3 it is more electronegative than H. And in case of same central atom, as the electronegativity of other atoms increases, bond angle decreases. Thus, bond angle in NF3 is smaller than that in NH3 because of the difference in the polarity of N in these molecules.

The structure of XeF6, is experimentally determined to be distorted octahedron. Its structure according to VSEPR theory is
octahedron
trigonal bipyramid
pentagonal bipyramid
tetragonal bipyramid
Two gases X (molecular weight MX) and Y (molecular weight My; My > Mx) are at the same temperature T in two different containers. Their root mean square velocities are Cx and Cy respectively. If the average kinetic energies per molecule of two gases X and Y are Ex and Ey respectively then which of the following relation(s) is(are) true?
Ex >Ey
Cx > Cy
Ex = Ey =(3/2)RT
Ex = Ey =(3/2)kBT
The IUPAC name of the following molecule is
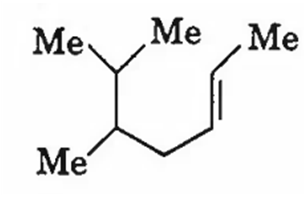
5,6-dimethylhept-2-ene
2,3-dimethylhept-5-ene
5,6-dimethylhept-3-ene
5-iso-propylhex-2-ene
