 Multiple Choice Questions
Multiple Choice QuestionsThe value of H for cooling 2 mole of an ideal monoatomic gas from 225C to 125C at constant pressure will be [ given Cp= ].
250 R
-500 R
500R
-250 R
The system that contains the maximum number of atoms is
4.25 g of NH3
8 g of O2
2g of H2
4g of He
The volume of ethyl alcohol ( density 1.15 g/cc) that has to be added to prepare 100 cc of 0.5 M ethyl alcohol solution in water is
1.15 cc
2 cc
2.15 cc
2.30 cc
The bond angle in NF3 (102.3) is smaller than NH4 (107.2). This is because of
large size of F compared to H
large size of N compared to F
opposite polarity of N in the two molecules
small size of H compared to N
The structure of XeF6, is experimentally determined to be distorted octahedron. Its structure according to VSEPR theory is
octahedron
trigonal bipyramid
pentagonal bipyramid
tetragonal bipyramid
Two gases X (molecular weight MX) and Y (molecular weight My; My > Mx) are at the same temperature T in two different containers. Their root mean square velocities are Cx and Cy respectively. If the average kinetic energies per molecule of two gases X and Y are Ex and Ey respectively then which of the following relation(s) is(are) true?
Ex >Ey
Cx > Cy
Ex = Ey =(3/2)RT
Ex = Ey =(3/2)kBT
For a spontaneous process, the correct statement(s) is (are)
B.
C.
For a process to be spontaneous
The IUPAC name of the following molecule is
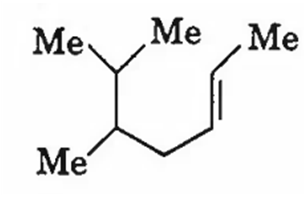
5,6-dimethylhept-2-ene
2,3-dimethylhept-5-ene
5,6-dimethylhept-3-ene
5-iso-propylhex-2-ene
