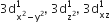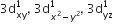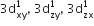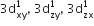Multiple Choice Questions
Multiple Choice QuestionsCopper sulphates dissolves in excess of KCN to give:
CuCN
[Cu(CN)4]3-
[Cu(CN)4]2-
[Cu(CN)4]2-
In which of the following pairs are both the ions coloured in aqueous solution?
(At. no.: Sc = 21, Ti = 22, Ni = 28, Cu = 29, Co = 2)
Ni2+, Ti3+
Sc3+, Ti3+
Sc3+,Co2+
Sc3+,Co2+
Al2O3 can be converted to anhydrous AlCl3 by heating:
Al2O3 with HCl gas
Al2O3 with NaCl in solid state
a mixture of Al2O3 and carbon in dry Cl2 gas
a mixture of Al2O3 and carbon in dry Cl2 gas
The appearance of colour is solid alkali metal halides is generally due to:
F- centres
Schottky defect
Frenkel defect
Frenkel defect
If  the standard emf of the reaction:
the standard emf of the reaction:
Fe + 2 Fe3+ →3Fe2+
will be:
0.330 V
1.653 V
For the reaction
2A + B → 3C + D
Which of the following does not express the reaction rate?

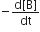


Consider the reaction
N2 (g) + 3 H2 (g) → 2 NH3 (g)
The equality relationship between  is:
is:




Which of the following is not chiral
2-butanol
2,3 -dibromo pentane
3- bromopentane
3- bromopentane
[Co(NH3)4(NO2)2]Cl exhibits:
linkage isomerism, ionisation isomerism and optical isomerism
Linkage isomerism, ionisation isomerism and geometrical isomerism
ionization isomerism, geometrical isomerism and optical isomerism
ionization isomerism, geometrical isomerism and optical isomerism
B.
Linkage isomerism, ionisation isomerism and geometrical isomerism
The compound [Co(NH3)4((NO2)2]Cl exhibits linkage, ionisation and geometrical isomerism.
Hence, its linkage isomers are
(i) [Co(NH3)2(NO2)2]Cl and [Co(NH3)4(ONO)2]Cl
(ii) Its ionisation isomers are
[Co(NH3)4 (NO2)Cl]NO2 and [Co(NH3)4(NO2)2]Cl
(iii) Its geometrical isomers are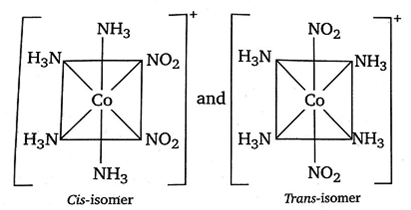
[Cr(H2O)6]Cl3 (at. no. of Cr = 24) has a magnetic moment of 3.83 BM, the correct distribution of 3d electrons in the chromium of the complex is: