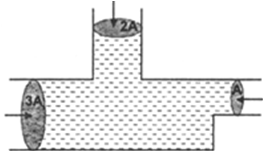
Thrust: The force exerted by liquid normal to any surface in contact with it, is called thrust.
Pressure: The thrust exerted by a liquid per unit area of surface in contact with liquid is called pressure.
The SI unit of pressure is N/m2 or Pascal (Pa).
Application of pressure:
i) Hydraulic machines
ii) Blood pressure - Pressure exerted by the circulating blood upon the walls of the blood vessels.
iii) Variation of pressure with depth - submarines are designed to withstand such enormous pressure.