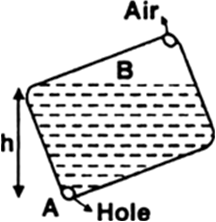
In a dropper, the liquid does not come out unless its bulb is pressed.
The pressure due to the height column of liquid inside the dropper is less than atmospheric pressure. That is the liquid is held inside the dropper against the atmospheric pressure. On pressing the dropper, the pressure inside is increased and when the pressure inside exceeds the atmospheric pressure, liquid comes out from the dropper.
174 Views