 Short Answer Type
Short Answer TypeCalculate the emf of the following cell at 25°C:

Given E°cell = + 0.46 V and log 10n = n
The rate of a reaction becomes four times when the temperature changes from 293 K to 313 K. Calculate the energy of activation (Ea) of the reaction assuming that it does not change with temperature.
[R = 8·314 J K-1 mol-1, log 4 = 0·6021]
For the complex [NiCl4]2-, write
(i) The IUPAC name.
(ii) The hybridization type.
(iii) The shape of the complex.
(Atomic no. of Ni = 28)
[NiCl4]2-
(i) IUPAC name = Tetrachloronickelate (II) ion
(ii) Hybridization of Ni in the complex [NiCl4]2- is sp3. The hybridisation scheme is shown in the following diagram.
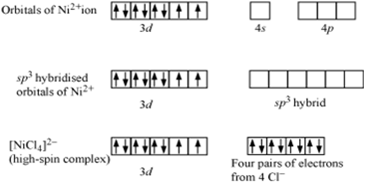
Hence the hybridisation of Ni2+ in the complex is sp3
(iii) As the hybridization of Ni is sp3 so, shape of the complex is tetrahedral.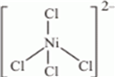
What is meant by crystal field splitting energy? On the basis of crystal field theory, write the electronic configuration of d4 in terms of t2g and eg in an octahedral field when
(i)![]() 0 > P
0 > P
(ii) ![]() 0 < P
0 < P
Give reasons for the following:
(i) Oxygen is a gas but sulphur is solid.
(ii) O3 acts as a powerful oxidising agent.
(iii) BiH3 is the strongest reducing agent amongst all the hydrides of Group 15 elements.
