 Multiple Choice Questions
Multiple Choice QuestionsThe 4th higher homologue of ethane is
butane
pentane
hexane
heptane
C.
hexane
The molecular formula of ethane is C2H6. In order to get its fourth homologue, we have to add 4 × CH2 = C4H8 to it.
C2H6 + C4H8 = C6H14
The molecular formula of hexane is C6H14. Hence, hexane is the fourth homologue of ethane.
The order of decreasing ease of abstraction of hydrogen atoms in the following molecule
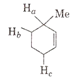
Ha > Hb > Hc
Ha > Hc > Hb
Hb > Ha > Hc
Hc > Hb > Ha
Among the following statements about the molecules X and Y, the one(s) which correct is (are)
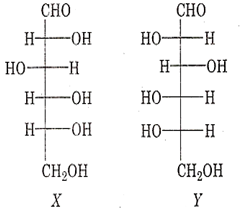
X and Y are diastereomers
X and Y are enantiomers
X and Y are both aldohexoses
X is a D-sugar and Y is an L-sugar
The electronic configuration of Cu is
[Ne] 3s2, 3p6, 3d9, 4s2
[Ne] 3s2, 3p6, 3d10, 4s1
[Ne] 3s2, 3p6, 3d3, 3d9, 4s2, 4p6
[Ne] 3s2, 3p6, 3d5, 4s2, 4p4
The rate of a certain reaction is given by, rate = k [H+]n. The rate increases 100 times when the pH changes from 3 to 1. The order (n) of the reaction is
2
0
1
1.5
The correct statement regarding the following energy diagrams is
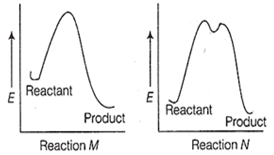
Reaction M is faster and less exothermic than reaction N
Reaction M is slower and less exothermic than reaction N
Reaction M is faster and more exothermic than reaction N
Reaction M is slower and more exothermic than reaction N
