 Multiple Choice Questions
Multiple Choice QuestionsA metal has a fcc lattice. The edge length of the unit cell is 404 pm. The density of the metal is 2.72 g cm3. The molar mass of the metal is (NA vogadro's constant = 6.02x 1023 mol-1).
40 g mol-1
30 g mol-1
27g mol-1
20 g mol-1
A magnetic moment of 1.73 BM will be shown by one among the following
[Cu(NH3)4]2+
[Ni(CN)4]2-
TiCl4
[CoCl6]4-
Roasting of sulphides gives the gas X as a by-product. This is a colourless gas with choking smell of burnt sulphur. and causes great damage to respiratory organs as a result of acid rain. Its aqueous solution is acidic acts as a reducing agent and its acid has never been insolated. The gas X is
H2S
SO2
CO2
SO3
Which is the strongest acid in the following?
H2SO4
HClO3
HClO4
H2SO3
C.
HClO4
The strength of oxyacids can also be decided with the help of the oxidation number central atom. Higher the oxidation number of central atom, more acidic is the oxyacid.
Since, in HClO4, oxidation number of Cl is highest, so HClO4 is the strongest acid among the given.
Which ofthe following lanthanoid ions is diamagnetic?
(At. nos. Ce = 58, Sm = 62, Eu = 63, Yb= 70)
Ce2+
Sm2+
Eu2+
Yb2+
Structure of the compound whose IUPAC name is 3-ethyl-2-hydroxy-4-methylhex-3-en-5-ynoic acid is
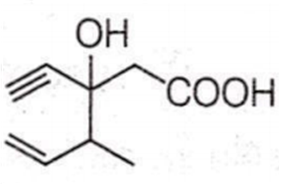
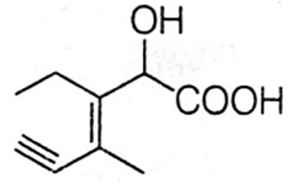
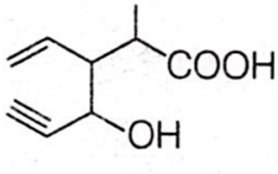
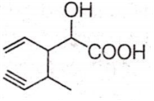
Antiseptics and disinfectants either kill or prevent growth of microorganisms. Identify which of the following is not true.
A 0.2% solution of phenol is an antiseptic while 1% solution acts as a disinfectant.
Chlorine and iodine are used as strong disinfectants.
Dilute solutions of boric acid and hydrogen, peroxide are strong antiseptics.
Disinfectants harm the living tissues.
