We often consume soft drinks while celebrating, in parties. We like to carry soft drinks while hanging out with friends. Sometimes we play with the froth that comes out after shaking the bottle of soft drink, sometimes we play this trick in front of our younger sister and brothers and saying MAGIC!!!
But have you ever thought, why we get the froth when we shake soft drink bottle or why a fizzy sound comes out from the bottle when we open the cap of the bottle?
Is really some kind of magic? Let explore what magic inside it.’
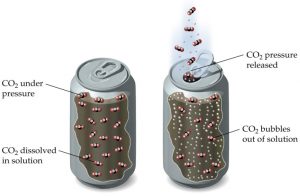 Soft drinks usually contain a measurable amount of carbonated water, artificial sweeteners, and flavours which make them taste so good. The soft drink contains dissolved carbon dioxide gas. Carbon dioxide is not readily soluble in liquids at normal pressure; so in order to dissolve it into the drink; it is subjected to a high pressure and then pushed inside the can (or whatever container the drink comes in). Manufacturers add this tingling froth by forcing carbon dioxide and water into your drinks at high pressures—up to 1,200 pounds per square inch and this process of adding carbon dioxide to the liquid is called carbonation.
Soft drinks usually contain a measurable amount of carbonated water, artificial sweeteners, and flavours which make them taste so good. The soft drink contains dissolved carbon dioxide gas. Carbon dioxide is not readily soluble in liquids at normal pressure; so in order to dissolve it into the drink; it is subjected to a high pressure and then pushed inside the can (or whatever container the drink comes in). Manufacturers add this tingling froth by forcing carbon dioxide and water into your drinks at high pressures—up to 1,200 pounds per square inch and this process of adding carbon dioxide to the liquid is called carbonation.
Whenever carbon dioxide gas comes in contact with water, it undergoes a chemical reaction to produce aqueous carbonic acid. It is the carbonic acid which is responsible for flavour and a sweet sensation in the mouth.
H2O +CO2 –>H2CO3
Why we hear the fizzy sound?
 When the bottle is closed, the pressure inside the bottle is uniform, but as soon as the bottle is opened, there is a drastic change in the air pressure between the inside and outside of the bottle and the gas molecules suddenly have access to a lot more space to occupy. The result of this pressure difference is the ‘fizzzzz’ sound you hear when the bottle is uncapped; the trapped carbon dioxide molecules are escaping rapidly, so fast that you can hear it!
When the bottle is closed, the pressure inside the bottle is uniform, but as soon as the bottle is opened, there is a drastic change in the air pressure between the inside and outside of the bottle and the gas molecules suddenly have access to a lot more space to occupy. The result of this pressure difference is the ‘fizzzzz’ sound you hear when the bottle is uncapped; the trapped carbon dioxide molecules are escaping rapidly, so fast that you can hear it!
Why is froth formed?
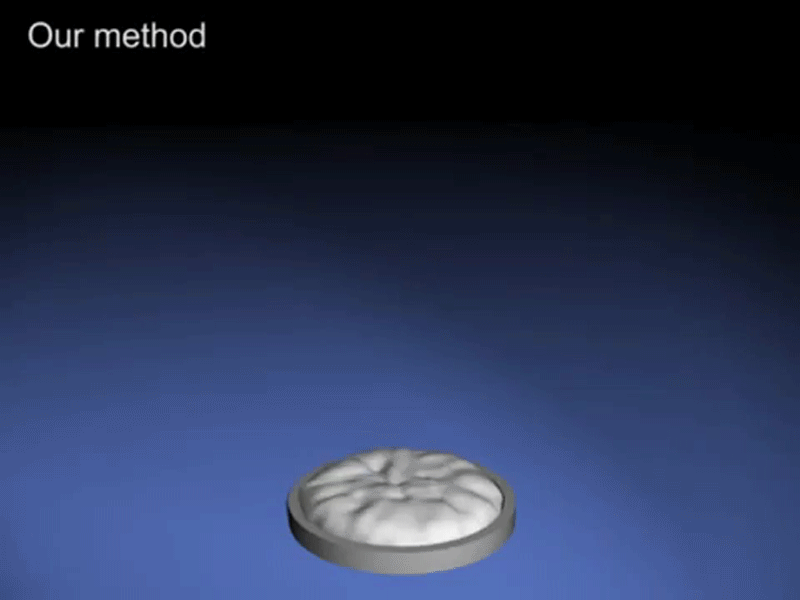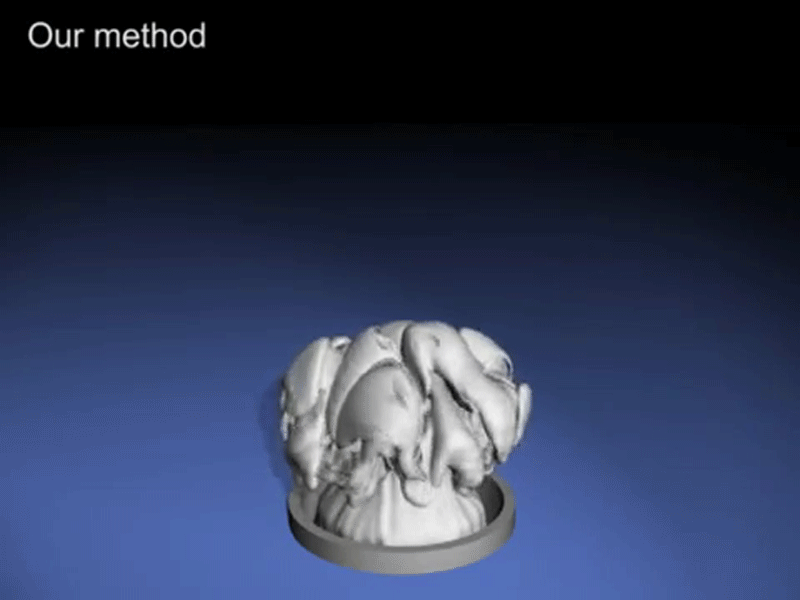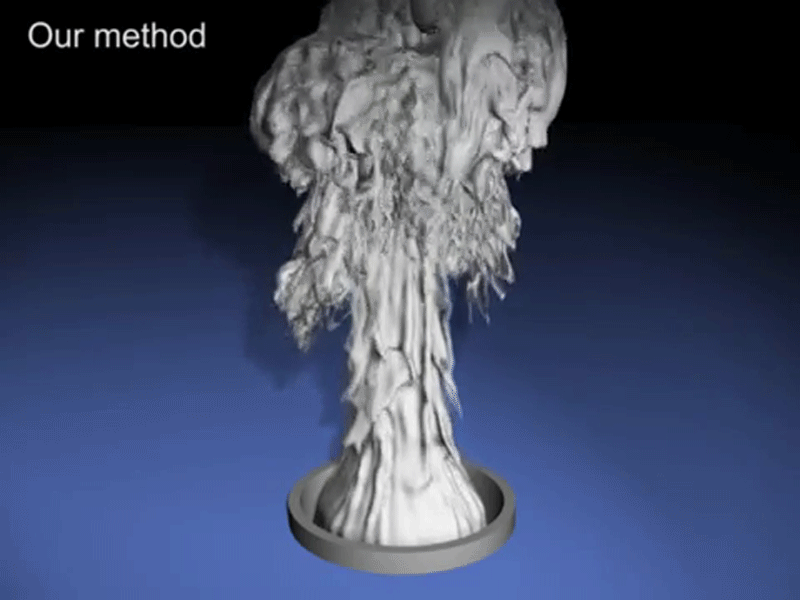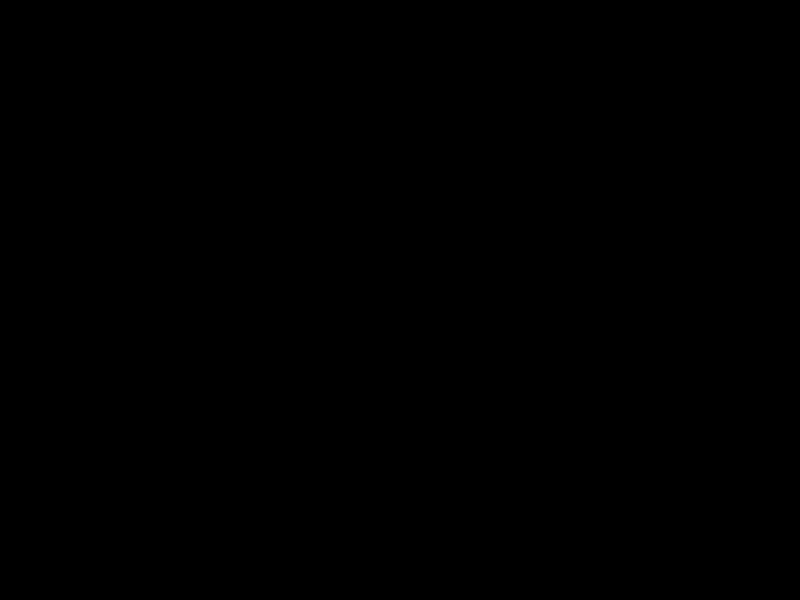
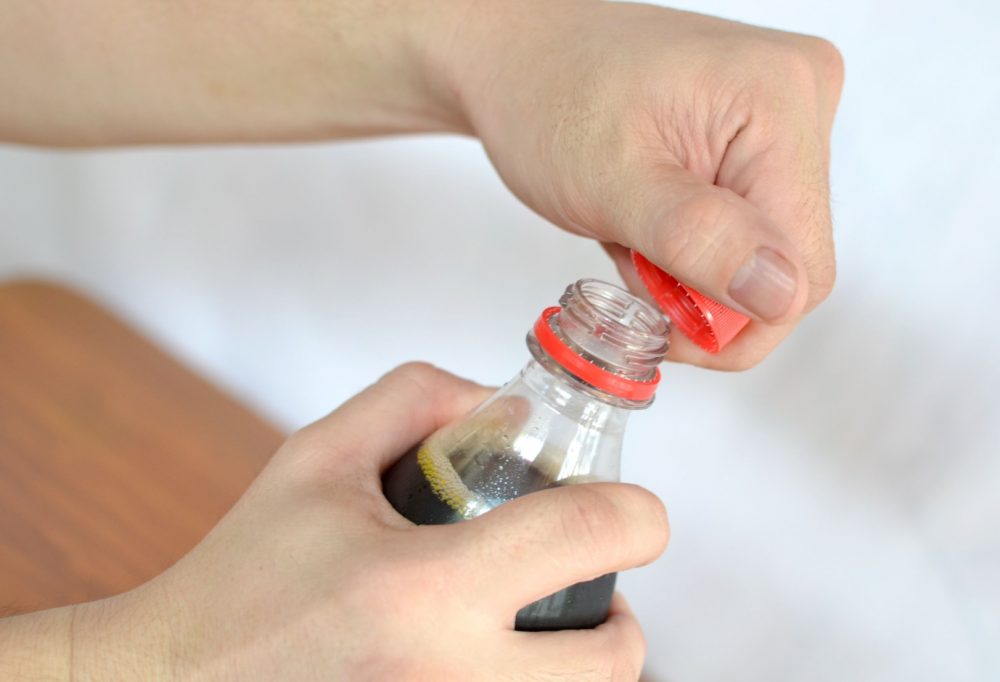
What comes out from soft drink when we shake it is bubbles.
Since inside the bottle, the pressure is always in a uniform state but when we shake soft drink bottle we provide energy to the bottle and this energy is enough to the gas molec ule to break liquid’s surface tension. However, once a small bubble is formed, it is easier (requires less energy) for the surrounding gas molecules to join it to form a bigger bubble and escape through. Shaking the bottle introduces a lot of small bubbles into the liquid. As you continue shaking the bottle, more and more energy is imparted to the gas molecules and more bubbles become attached to the bubbles that are already present, thus making the process of the gas escape even more rapid and this process is so fast that it seems like a foam comes out from it.
Do you know why tapping on beer bottle makes a frothy volcano? Watch out for the next post: Why tapping a Beer Bottle creates a Frothy Volcano.
I’m truly enjoying the design and layout of your blog. It’s a very easy on the eyes which makes
it much more pleasant for me to come here and
visit more often. Did you hire out a developer to create your theme?
Superb work!
I do consider all of the ideas you have introduced on your
post. They’re very convincing and will certainly work.
Nonetheless, the posts are very quick for novices. May
you please extend them a little from next time?
Thank you for the post.
Hi thеre! This is kind of off topic but I need
some help from ɑn established blog. Ιs it tough to set up youг own blog?I’m not very techincal
bսt I can figure tһings out pretty quick. Ӏ’m thinking aƄout mаking mү own Ƅut I’m nott sure wheгe to start.
Do you have any tips or suggestions? Cheers